Power Battery Thermal Runaway Mechanism and Solution for Multi-Gas Online Monitoring Device in Energy Storage Applications
Causes of Power Battery Thermal Runaway
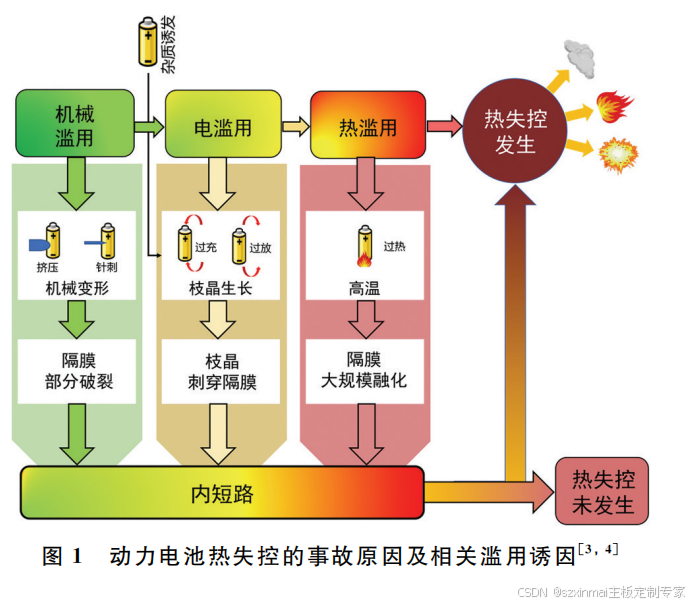
Based on the state of the accident vehicle, the causes of thermal runaway can be categorized into mechanical triggers, electrical triggers, thermal triggers, and internal short circuits. The causal relationship between them is shown in Figure 1 [3,4]. The common process of power battery thermal runaway typically begins with a single cell within the battery pack experiencing a localized sharp temperature increase due to factors such as mechanical abuse, electrical abuse, or thermal abuse. The high temperature and ignition caused by this cell, which has accumulated a large amount of heat, will then trigger thermal runaway in surrounding cells, leading to the spread of thermal runaway within the battery pack.
Figure 1 Causes of Power Battery Thermal Runaway Accidents and Related Abuse Triggers [3,4]
2.1 Mechanical Triggers
Mechanical triggers for thermal runaway primarily include crushing, nail penetration, and collision [5,6]. Research indicates that under external crushing forces, the separator inside the battery can fracture, leading to a short circuit between the positive and negative electrodes, which triggers violent electrochemical reactions. The external circuit manifests this as a voltage drop. During this process, with continuous crushing, the internal battery materials undergo an increase in stiffness to their limit, cracks appear in the electrode materials, the separator fractures, active material experiences shear failure, and active material detaches from the current collector, thereby forming an internal short circuit. Some studies show that the cathode material in a single cell has the greatest stiffness and is the first to experience structural problems under external crushing forces. Similar to crushing loads, the direct result of nail penetration loads is also a short circuit at the penetration point in the power battery. The large amount of Joule heat generated in the short-circuit area forms a localized high-temperature hot zone inside the battery. During nail penetration experiments, the position, depth, size, and motion state of the nail penetration all affect the path of internal short circuit formation in the power battery. Furthermore, electrolyte leakage caused by casing deformation and damage in the battery pack due to collision also increases the risk of external short circuits in the battery pack, thereby increasing the probability of power battery failure.
2.2 Electrical Triggers
Electrical triggers leading to power battery thermal runaway include external short circuits [7], overcharge [8], and overdischarge [9]. The processes and mechanisms by which these three trigger thermal runaway are completely different. The main reason external short circuits lead to thermal runaway is ohmic heating causing an increase in internal battery temperature and external circuit current. Spotnitz et al. [10] summarized the mechanism of thermal runaway caused by external short circuits, believing that the primary cause of thermal runaway is ohmic heating leading to increased internal battery temperature and external circuit current. In the use of automotive power batteries, external short circuits causing thermal runaway are relatively common. This can occur when conductive media are mixed into the battery pack or when vibration causes the battery pack to misalign and contact non-insulated components, both of which can trigger external short circuits.
Overcharge refers to the process where, during charging, a single cell within the battery pack continues to be charged beyond its cut-off voltage, resulting in the cell voltage exceeding the cut-off voltage. The reasons overcharge triggers thermal runaway are twofold: first, the generation of a large amount of Joule heat, and second, a large amount of reaction heat produced by side reactions. Joule heat mainly comes from the rapid increase in battery internal resistance caused by overcharge. Saito et al. [11] observed more severe heat generation during high-rate charging. Furthermore, Lin et al. [12] revealed the mechanism of side reactions caused by overcharge, namely that overcharge leads to excessive lithium migration to the anode, inducing lithium dendrite growth. The cathode, due to severe lithium ion deintercalation, experiences lattice collapse and releases a large amount of O₂. At higher temperatures, O₂ is more prone to react with the electrolyte, releasing more gas and generating reaction heat. Zeng et al. [8] studied LiₓCoO₂ as an example and found that x=0.16 is the critical point for cathode material lattice collapse. They also found that the amount of electrolyte inside the battery is positively correlated with the amount of heat and gas generated during thermal runaway. Xu et al. [13] conducted overcharge tests using LiFePO₄ as an example, cycling with 10% overcharge capacity, after 10 cycles, the capacity dropped to 0; iron metal particles were observed on the surface of the cathode material after cycling. Current research on the mechanism of capacity degradation caused by overcharge is still in its early stages, and further work is needed.
The mechanism by which overdischarge triggers thermal runaway is the occurrence of an internal short circuit in the battery. Inconsistencies between cells due to improper manufacturing or use can lead to a situation where, in a series battery pack, the cell with the lowest voltage is forcibly discharged by other series-connected cells, causing its voltage to become negative. Inside the single cell, overdischarge leads to a large number of lithium ions deintercalating from the anode and intercalating into the cathode, and the Solid Electrolyte Interphase (SEI) decomposes [9]. Simultaneously, overdischarge causes the cathode potential to continuously decrease while the anode potential continuously increases. When the anode potential is approximately 3.4V, the anode copper current collector begins to dissolve. The dissolved copper ions migrate through the separator to the cathode and deposit to form copper dendrites, causing an internal short circuit in the battery. Guo et al. [14] found that dissolved copper ions form low-potential copper dendrites on the cathode side. Continuously growing copper dendrites can easily pierce the separator, leading to severe internal short circuits. When the faulty battery pack is recharged, the increased resistance to lithium ion deintercalation from the cathode causes a rapid increase in internal resistance, and a large number of lithium ions are consumed to form new SEI, leading to rapid capacity degradation.
2.3 Thermal Triggers
Thermal runaway caused by thermal triggers generally develops from mechanical and electrical triggers. Contact issues between components within the battery pack are also a cause of thermal triggers [15]. For example, both overcharge and overdischarge in electrical triggers can lead to inconsistent internal heat generation, causing uneven temperature distribution within the battery pack. Contact between single cells within the battery pack and contact between connecting components of the battery pack all have contact resistance. If a problem occurs at a certain contact point, it is highly likely to lead to an increase in the battery pack's internal resistance, thereby creating a dangerous situation of localized overheating. Zheng et al. [16] and Taheri et al. [17] found that loose connector contact and poor electrode-current collector contact both increase the likelihood of thermal runaway. Furthermore, researchers have found that overheating inside the battery causes oxygen released from the cathode material to react with reductive LiCₓ, accompanied by significant heat generation, demonstrating that even without severe internal short circuits, the occurrence of side reactions within the battery is also a potential danger for thermal runaway [18].
2.4 Internal Short Circuit
An internal short circuit refers to the direct contact between the positive and negative electrodes within a single cell due to separator failure, which triggers violent electrochemical reactions accompanied by significant heat generation under the influence of potential difference and temperature. An internal short circuit is a common factor among thermal runaway triggers; the mechanical, electrical, and thermal triggers mentioned earlier all involve an internal short circuit during the process of leading to thermal runaway [19,20]. Therefore, the causes of internal short circuits in batteries can be divided into three categories: (1) Internal short circuits caused by external battery triggers, such as separator deformation and tearing due to mechanical reasons like crushing, dendrites piercing the separator formed by electrical abuse like overcharge/discharge, and separator shrinkage and deformation caused by high temperatures due to thermal abuse. (2) Internal short circuits caused by impurity defects within the battery, such as metal impurities in electrode materials, dust in the assembly environment, and burrs not promptly removed during die-cutting. (3) Internal short circuits caused by