NXP IMX8M + FPGA-based In Vitro Diagnostic POCT Device
In Vitro Diagnostic POCT Devices
With an aging population and increasing emphasis on health, POCT (Point-of-Care Testing), a sub-segment of IVD (In Vitro Diagnostics), continues to grow in market size. POCT (point-of-care testing) refers to clinical tests and bedside testing performed near the patient. It is a testing method that uses portable analytical instruments and corresponding reagents to quickly obtain test results. Common POCT devices include biochemical analyzers, immunoassay analyzers, PCR instruments, etc., employing various technologies such as dry chemistry, immunoassay, chemiluminescence, biosensors, biochips, and microfluidics. They are applied in fields such as cardiac markers, blood glucose, blood gas, electrolytes, coagulation, infectious diseases, pregnancy, drug testing, drunk driving detection, food safety, and tumor marker detection.

POCT Advantages
Speed: Compared to traditional diagnostics, POCT streamlines steps such as sample transportation and processing, eliminating the need for complex sample preparation. Test results can be obtained in just a few minutes or tens of minutes, significantly shortening diagnosis time and saving time for patient visits and emergency treatment.
Ease of Use: POCT has no specific requirements for operators. Medical staff or patients can use the instruments after reading the manual or undergoing simple training, making them popular in grassroots hospitals, outpatient clinics, and home settings.
Portability: POCT devices are generally compact, battery-powered, and highly portable. They are not restricted by time or location, can serve patients 24 hours a day, and provide convenience for applications in complex scenarios such as disaster relief sites and entry-exit quarantine.

Unlike the complexity of large laboratory testing instruments, POCT devices are typically compact and have integrated internal functions. With technological advancements, POCT instruments are evolving towards full automation, intelligence, and high throughput. POCT devices require simple procedures, high detection accuracy, the ability to continuously measure multiple samples and perform multiple analyses simultaneously, automatic data logging, automatic upload of test results, and self-diagnosis of faults. This places high demands on the device's main control board.
Hardware Recommendation
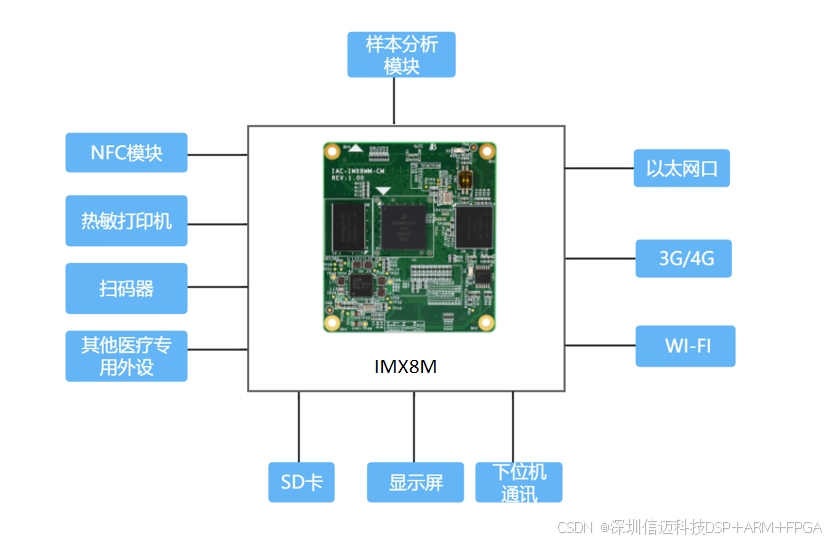
As the main control board for medical POCT instruments, Xinmai's IMX8MM core board is based on a quad-core Cortex-A53 platform. The core board is powered by an NXP i.MX8M mini processor featuring 4*Cortex-A53 + Cortex-M4 cores. It offers high performance and low power consumption, with a main frequency up to 1.8GHz. It can be equipped with large-capacity memory, providing sufficient hardware resources for medical devices.
It provides powerful multimedia capabilities, supporting various forms of hardware encoding and decoding, and a 4-channel MIPI high-definition display interface to meet the HMI display requirements of various medical devices.
It boasts rich interface resources, including dual Gigabit Ethernet ports, CAN, I2C, RS232, USB, and other data communication interfaces, to accommodate the connection of various POCT peripherals.
It supports wireless communication methods such as 3G/4G and Wi-Fi, enabling test results to be uploaded to the cloud. By interfacing with POCT management systems and hospital HIS/LIS systems, it facilitates remote test result analysis and remote diagnosis and treatment. It also supports device self-diagnosis, with operating status and fault alarms wirelessly uploaded to the management backend, enabling remote device maintenance and OTA remote upgrades.
It supports Linux and Android operating systems, offering open-source low-level code and technical support, which reduces development difficulty and cost, allowing customers to focus on upper-layer applications during secondary development.
Given the stringent hardware requirements for medical devices, an industrial-grade design can be chosen to ensure the stability of device operation. Advantages such as an ultra-long lifecycle further demonstrate that the core board is an ideal choice for the main control of POCT in vitro diagnostic devices.
The implementation of national policies such as hierarchical medical treatment and import substitution for medical devices, coupled with the boom in the medical market triggered by the COVID-19 pandemic, have brought unprecedented opportunities to the POCT market. In the future, with the further integration of technologies such as biochips, biosensors, and optoelectronic analysis into the medical field, POCT products will become increasingly precise and efficient in detection, and their application scenarios will become increasingly widespread.