i.MX8M Plus + FPGA + AI Patient Monitor Solution
A patient monitor is a device or system that measures and controls a patient's physiological parameters, compares them with known set values, and can issue an alarm if these values exceed the set limits.
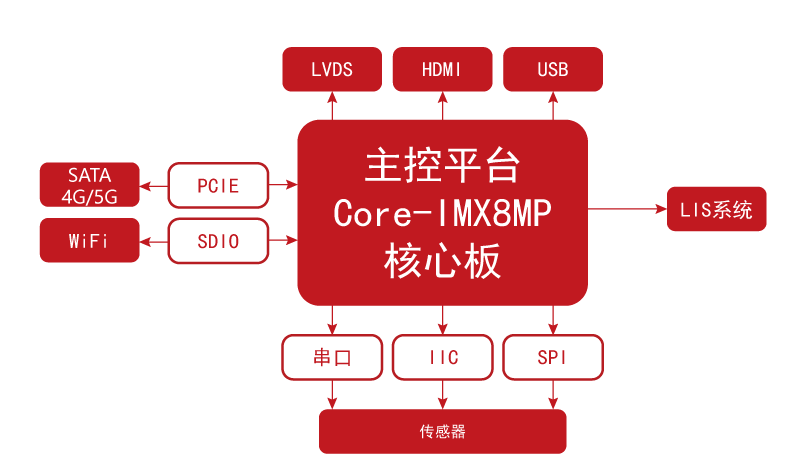
(1) Patient monitors primarily collect and measure human physiological parameters such as ECG, blood pressure, blood oxygen, and body temperature. This requires collecting and processing a large amount of data, and the system needs to handle multiple tasks in parallel, which consumes significant CPU resources. Based on the i.MX8M Plus, NXP's first i.MX8 series product to integrate a dedicated Neural Processing Unit (NPU), it focuses on machine learning and vision, advanced multimedia, and highly reliable industrial automation. It features powerful quad-core or dual-core Arm® Cortex®-A53 processors with an NPU, operating at up to 2.3 TOPS. The core board uses gold-plated edge connectors and integrates the CPU, LPDDR4, eMMC, PMIC, SPI FLASH, and dual Gigabit Ethernet PHYs, allowing users to simplify product design.
(2) A good human-machine interaction (HMI) experience is a fundamental requirement for device services. The i.MX8M Plus supports Linux 4.1.15 + Qt 5.6, Android 4.4.2, Android 6.0, and Linux 3.0.35 + Qt 4.8.5, facilitating various applications for engineers with different needs.
(3) Different application scenarios may require two screens to display simultaneously. A small screen can be used for human-machine interaction, while a large screen can serve as a duplicate or extended display. The i.MX6Q supports dual 8-bit LVDS, RGB, HDMI, and MIPI display interfaces, with resolutions up to 1080P. It also supports dual-screen simultaneous display and dual-screen extended display.
(4) A product with comprehensive functional interfaces can eliminate the hassle of various interface verifications. The i.MX8M Plus is a highly integrated application processor that supports various common interfaces such as network, serial port, USB, audio, SPI, and I2C, meeting the needs of diverse product applications.
(5) Certification and clinical testing for medical products take a long time, in addition to an approximately one-year development cycle. Therefore, the longevity of the processor's lifecycle is paramount. The i.MX8M Plus processor has a lifecycle of over 10 years, ensuring customers don't have to worry about chip obsolescence.
(6) Before medical diagnostic devices can be launched, they must pass stringent electromagnetic compatibility (EMC) tests. The results of electrostatic discharge (ESD), surge, and electromagnetic interference (EMI) tests directly impact the accuracy of diagnostic results. The i.MX8M Plus core board uses an immersion gold 12-layer PCB process to ensure optimal electrical characteristics.
Solution Topology Diagram