【RK3588/Sophon/Nvidia Smart Box】AI Algorithms Applied to Intelligent Monitoring of China National Biotec Group Vaccine Production, Empowering Production Safety and Enhancing Quality Control
Frequent Food and Drug Quality Incidents Due to Operational Errors
Computer Vision Detection Technology Offers New Approaches to Regulation
In recent years, various food and drug quality incidents caused by human operational errors have occurred frequently. For example, an employee failing to perform a dual verification when retrieving raw materials and confirming weight led to "medication mix-ups"; an employee failing to sterilize raw milk according to production steps resulted in cheese contamination; and an employee mistakenly mixing up two vaccine raw material components led to 15 million doses of vaccine being scrapped. All these incidents caused severe and irreversible consequences.
The requirements for environmental hygiene, process flow, and product quality in food and drug production are becoming increasingly stringent. As computer vision detection technology continues to mature, it can not only detect product defects, packaging label information defects, etc., but also identify whether the sequence of actions is compliant, whether actions are standard, or whether actions are omitted, significantly improving detection efficiency and offering advantages that traditional manual visual inspection lacks. It provides new regulatory approaches for risk identification and quality control in food and drug production processes.

△Image source: Internet
China National Biotec Group (CNBG)
Machine Vision-Based Intelligent Monitoring of Vaccine Production Process
China National Biotec Group (CNBG) is a comprehensive biopharmaceutical enterprise with a long history, a complete product portfolio, and large scale, integrating scientific research, production, sales, and postgraduate education. To enhance the automation level of vaccine production process supervision, CNBG has introduced computer vision technology on the basis of its existing video surveillance system to build an intelligent monitoring and prevention system, aiming to leverage artificial intelligence to strengthen risk identification and timely correction for operational compliance, biosafety, sterility requirements, and other Good Manufacturing Practice (GMP) risks during vaccine production.
In CNBG's "Machine Vision-Based AI Monitoring of Vaccine Production Process" project, targeting two major scenarios: "Virus Inactivator Addition" and "Strain Retrieval", 17 types of AI vision algorithms for vaccine production SOP monitoring were custom-developed. These algorithms monitor the compliance of personnel operational behavior during vaccine production, including whether the sequence of actions is compliant, whether actions are standard, or whether actions are omitted. This is all managed uniformly through an AI monitoring platform, empowering production safety and enhancing quality control.
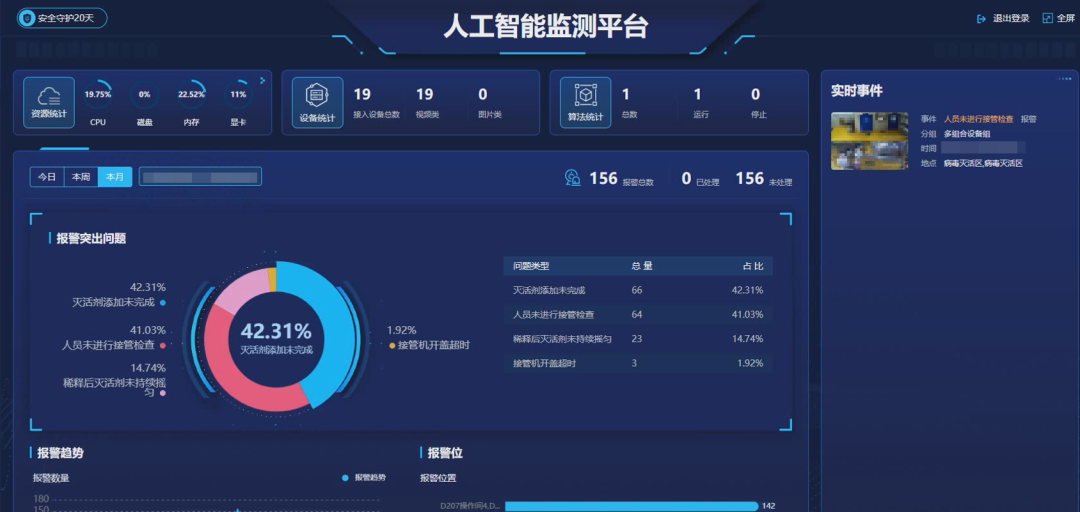
△CNBG AI Monitoring Platform for Vaccine Production Process
1
Virus Inactivator Addition Scenario
As the only material addition operation in the virus culture inactivation area, monitoring personnel need to track the flow and usage of the inactivator to confirm the compliance of the addition process and ensure the effectiveness of virus inactivation.
Eight algorithm models were integrated to form a single algorithm for regulating the entire operational process in this scenario, including V-sign gesture recognition, protective suit recognition, mask recognition, positive pressure mask recognition, tubing machine switch status recognition, tubing connection inspection recognition, isolation valve switch status recognition, and culture medium shaking recognition. This achieves standardized regulation of the inactivator addition process in vaccine production.

△Schematic Diagram of V-Sign Gesture Recognition Algorithm
2
Strain Retrieval Scenario
As a critical material for controlling biopharmaceutical production, the safety of its storage method and the compliance of its retrieval process have a crucial impact on product quality.
Thirteen algorithm models were integrated to form a single algorithm for regulating the entire operational process in this scenario, including V-sign gesture recognition, protective suit recognition, mask recognition, positive pressure mask recognition, dual-person dual-lock recognition, refrigerator door switch status recognition, transfer box switch status recognition, disinfection recognition for holding strain vials, anti-freeze glove recognition, strain vial counting recognition, logbook recognition, refrigerator locking recognition, and personnel circling recognition. This achieves standardized regulation of the strain retrieval process in vaccine production.
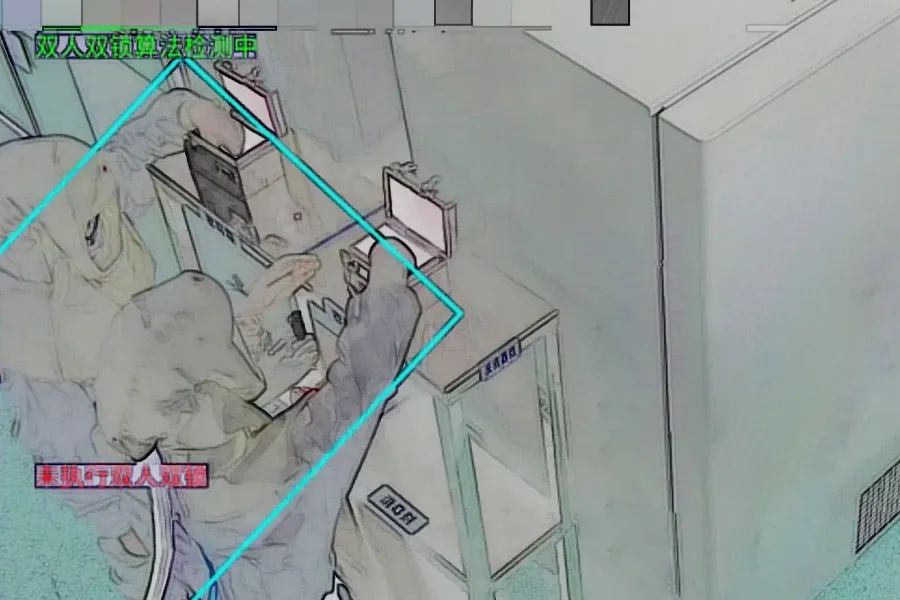
△Schematic Diagram of Dual-Person Dual-Lock Recognition Algorithm
During the algorithm development process for this project, key technologies such as object detection, object tracking, and semantic segmentation were utilized and applied to control operational compliance in multiple stages of vaccine production. Furthermore, the algorithms are deeply integrated with domestic Xinchuang (information technology application innovation) products and technologies, providing greater scope for future development.
China NMPA and Others Highly Affirm Project Value
Enormous Potential for Intelligent Regulation in Pharmaceutical Production
AI empowers safe production of pharmaceuticals and vaccines, transforming passive "supervision" into proactive "monitoring" by enabling real-time pre-event warnings, continuous in-event monitoring, and standardized post-event management. This achieves standardized workshop operations, controllable personnel behavior, and tiered decision analysis, truly realizing informatized management of the vaccine production process.
Previously, the acceptance meeting for the "Machine Vision-Based AI Monitoring of Vaccine Production Process Project" was held in Beijing. Experts and leaders from China NMPA (National Medical Products Administration), China National Biotec Group (CNBG), Wuhan Institute of Biological Products, etc., attended and highly affirmed the achievements and value of the project.
Based on the immense potential of computer vision detection technology in regulating standard actions and processes during vaccine production, it will be applied to more scenarios in pharmaceutical enterprises' production, developing more high-quality and practical AI vision algorithms, and driving the intelligent development of the pharmaceutical industry.